Understanding mechanical signalling in the microcirculation
A layer of proteins lining our blood vessels called the Endothelial Glycocalyx Layer (EGL) protects the vessel walls from potentially harmful levels of fluid shear stress. However, the EGL must also allow the vessel walls to receive mechanical information about the flow of blood in the vessel in order to trigger biochemical events.
Blood vessels are lined with a brush-like layer, called the Endothelial Glycocalyx Layer (EGL), which is a complex structure of hydrated membrane-bound proteins, interwoven with glycosaminoglycans. This layer is believed to serve a number of functions in the microvasculature, including protection of the vessel walls from potentially harmful levels of fluid shear stress. However, the ability of the EGL to attenuate fluid shear stresses is seemingly at odds with the need for the vessel walls to receive mechanical information about the flow of blood in the vessel, which acts as a trigger for certain biochemical events. A current hypothesis is that much of the mechanical load is carried through the solid portion of the EGL, rather than through its hydrating fluid. However, the fragile nature of the EGL has made this theory difficult to conclusively prove this idea in experiments.
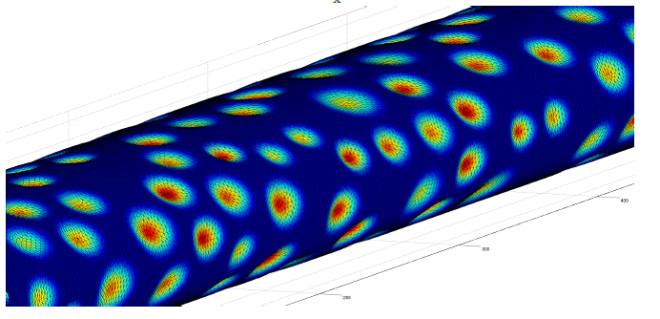
PhD students Pavel Sumets and Tet Chuan Lee investigated this idea, under the supervision of Dr Richard Clarke, Dr John Cater and Dr David Long at the Department of Engineering Science, University of Auckland. Since modelling the microstructure of the EGL in all its complexity is computationally infeasible, they adopted a volume-averaged macroscopic level description, using Biphasic Mixture Theory. In an attempt to represent physiological reality as closely as possible, the geometry that they have used in the simulation was informed by actual biological data obtained from colleagues Dr Jennifer Bodkin and Prof. Sussan Nourshargh at the Centre for Microvascular Research, William Harvey Research Institute, Barts and The London Medical School, Queen Mary University of London.
In order to facilitate efficient computation, the first boundary-integral representation of the governing Biphasic Mixture Theory equations were derived [1]. This means that the poroelastohydrodynamics of the EGL can be represented by quantities defined only on the surfaces of the geometry (rather than throughout its entire volume). Essentially this reduces the dimensionality of the computational problem.
“Even with these savings,” says Dr Richard Clarke, “The computational challenges were significant, and required the high performance computing facilities available through NeSI. For simulations using an idealised two-dimensional geometry [1], construction of the computational system was distributed across 96 cores, each requiring 1.5Gb of memory, with a total solve time of 600 CPU hours for each of the 10 physiological scenarios considered. For the three-dimensional simulations using the biologically informed geometries [2], each simulation required up to 30,000 CPU hours at the highest resolution.”
This computational study will now be extended to incorporate some of the non-homogeneities associated with the EGL's geometry, as well as electromechanical effects. There is some debate around the extent to which these effects are important in allowing the EGL to recover its shape after being crushed by passing blood cells, as compared with elastic restoring effects. It is hoped that future computational studies will be able to help shed some light on this issue.
References
[1] P. P. Sumets, J. E. Cater, D. S. Long, R. J. Clarke. (2015) A boundary-integral representation for biphasic mixture theory, with application to the post-capillary glycocalyx. Proc. Royal Soc. Lond. A. 471
[2] T.C. Lee, D.S. Long and R.J. Clarke (2015) Effect of Endothelial Glycocalyx Layer redistribution upon microvessel poroelastohydrodynamics. (Submitted)