Finding genetic help for Māori health
Dr Tony Merriman is a research professor at the University of Otago. There, he is studying the genes of New Zealand and Cook Island Māori to understand why Māori people have one of the highest rates of gout and hyperuricemia in the world.
“Our current project maps 130 genomes of Māori people. Ancestrally, Māori are Eastern Polynesian and we have very little information on the background genetic structure of those populations. We need that information to help us do our research,” Tony said.
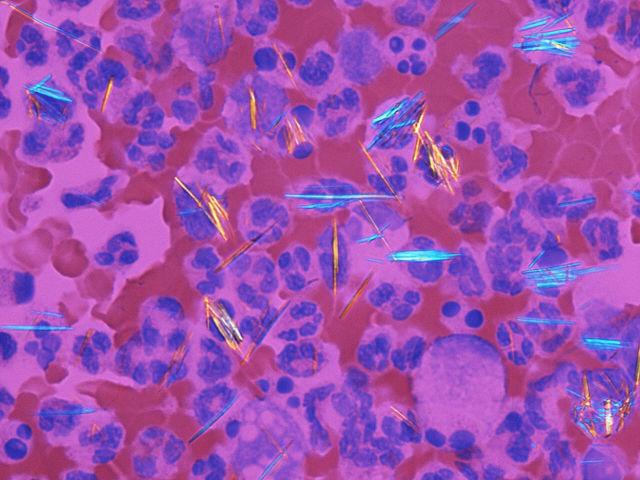
Hyperuricemia is a condition where too much urate is in the blood. Over time, this urate can crystallise, leading to a number of painful and potentially fatal diseases, including gout.
“With genetics research we may be able to identify genes causing hyperuricemia or gout itself. The pharmaceutical industry can assess whether those genes are valid targets for new drugs or repurposed existing drugs.”
A common trigger of gout attacks is diet – particularly purine-rich foods such as alcohol, meat and seafood. However, this doesn’t explain the high frequency of sufferers among Māori people – which can be up to double the rate of European-ancestry New Zealanders.
“We know it goes back to pre-colonisation, so it’s not just our modern diet,” Tony said. “One theory is having elevated urate levels was a defence mechanism against malaria. But we don’t really know. You can come up with ideas but they’re difficult to test.”
The genetics research for these drugs relies on mapping human genomes. A genome is the total set of genetic information within each human – similar to a blueprint for that person. Each genome is unique, but people with a shared ancestry will have more genetic information in common than those more distantly related.
This genetic variation becomes important when designing medicine because differences in genes can change how the medicine reacts to a person. A medicine which helps one person may harm another. Most medicines are designed for people of European ancestry, which can make treatment of people with different ancestries, such as Māori people, difficult.
“There are hundreds of thousands of genomes mapped for Europeans. There are less than two hundred genomes of Māori people that have ever been done in a medical context. What we’re aiming for is to have equivalent research done as to other populations,” Tony said.
It is difficult to compare exact numbers of genomes mapped for European and Māori ancestries due to a lack of data. However, there is a widely-acknowledged bias towards European genome mapping in medical research. Tony’s team are hoping to counteract this bias.
The sequencing of these genomes is done in Australia, at the Garvan Institute of Medical Research. There, DNA is broken up into small snippets and read by a gene sequencer, before being sent back to Tony’s team to reassemble using NeSI’s Mahuika supercomputer.
Finally, the mapped genomes are compared to what is called a reference genome. Comparison helps Tony’s team spot differences between Māori and other ancestries’ DNA. Once they’ve compared the genomes, the next step is to find out what those different segments control in the human body.
“We want to figure out what the genetic variants do in a biological context. What are they doing in the body? We’re going to start moving more into functional work: what and when they’re expressed; how the variants regulate biological process. Interpreting them is challenging.”
For many Māori people, gout is a painful inheritance. It takes close cooperation with Māori communities, passionate researchers like Tony and resources like NeSI’s Mahuika supercomputer to provide better treatment to those at risk. Working together may help improve the lives of many people.